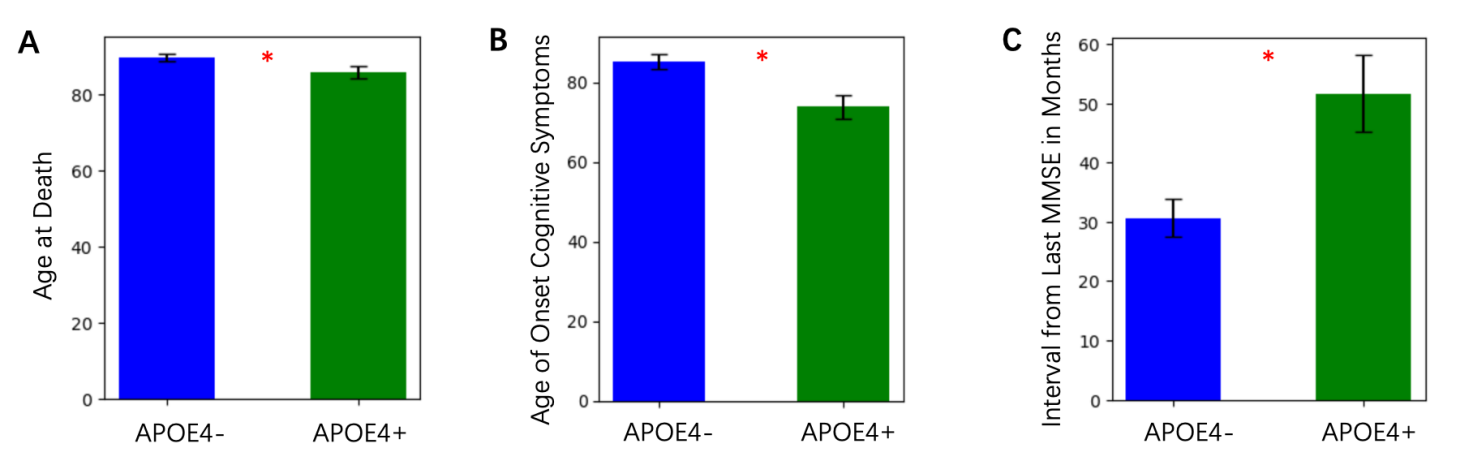
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder characterized by cognitive decline, amyloid plaque accumulation, and tau pathology. The Apolipoprotein E4 (ApoE4) allele is the strongest genetic risk factor for late-onset AD, contributing to increased amyloid burden, neuroinflammation, and brain atrophy. However, the cellular and structural mechanisms underlying ApoE4’s impact on disease progression remain incompletely understood. In this paper, we analyzed multi-modal datasets from the Seattle Alzheimer’s Disease Brain Cell Atlas (SEA-AD) to investigate differences between ApoE4+ and ApoE4- individuals. 116 parameters were assessed, including neuropathology, imaging, and clinical outcomes and we identified 15 key parameters that are more significant and representative between the groups. ApoE4+ individuals exhibited earlier cognitive decline, greater tau and amyloid pathology, and accelerated brain atrophy, particularly in cortical and subcortical regions. RNA sequencing of the Prefrontal Cortex (PFC) and Middle Temporal Gyrus (MTG) revealed significant reductions in Sst Chodl, Sncg, and Vip interneurons in ApoE4+ individuals. These findings suggest that ApoE4 carriers experience widespread neurodegenerative changes, including altered inhibitory neuron composition, which may contribute to disease progression. Understanding how ApoE4 affects inhibitory networks could provide new insights into Alzheimer’s pathology and potential therapeutic targets.


